Jan 7, 2013 | Newsletters
Click Chemistry has become a powerful method for DNA modification. Once an alkyne or azide moiety is incorporated into DNA, it can subsequently be conjugated by a simple CLICK reaction[1] with a reporter molecule such as biotin or a fluorescent dye.
A very prominent version of CLICK is the so-called “Cu(I)-free [2+3] cycloaddition strategy” that is based on strained cyclooctynes. Their use decreases the activation energy for the CLICK reaction, enabling it to be carried out at low temperatures, without the need for catalysis, and with greater efficiency than conventional CLICK[2-7].
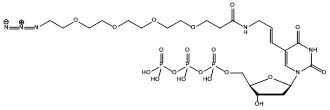
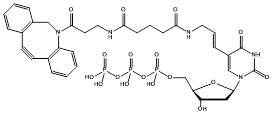
We report here a universal DNA labeling strategy that relies on efficient PCR-incorporation of the CLICKable dUTP analogs Azide-PEG4-aminoallyl-dUTP (Figure 1; JBS Cat.No.: NU-1705), and 5-DBCO-dUTP (Figure 2; JBS Cat.No.: CLK-T09) into a DNA fragment.